 
Pitting can penetrate deep into the tubing walls, creating a situation where tubing could fail 3. The consequences are accelerated pitting, perforation, leaks and collapse of material structures and equipment’s. The corrosive nature of the marine environment has a strong influence on the passivation characteristics of the steel.Once the passive film weakens and breakdown, an electrochemical cell initiates whereby the substrate Iron oxidizes to iron oxide. The corrosion resistance and electrochemical behaviour of stainless steels in seawater is of great interest due to their extensive application therein. Petrochemical structures routinely apply stainless steels tubing in process instrumentation and sensing, chemical inhibition, hydraulic lines, impulse lines, and utility applications, over an extensive variation of temperatures, flows, and pressures. Cost of corrosion damage in the maritime industry as a result of the routine exposure of vessels and marine structures to these chloride containing environments has increased geometrically every year with an estimated total cost of between $50-80 billion worldwide with the oil and gas taking a significant chunk of the cost 1, 2. The major cause of corrosion in these environments is the presence of chloride anions. The basic cause of metallic corrosion is the inherent instability of metal alloys in their refined forms as they tend to revert to their natural states through the processes of corrosion. Corrosion problems in these environments have been researched into but failures still occur. Seawater environments are extensively used by industries such as shipping, offshore oil and gas production, power plants and coastal industrial plants mainly for cooling, oil field water injection and for desalination plants. 
Study of the Corrosion Resistance of Type 304L and 316 Austenitic Stainless Steels in Acid Chloride Solution. The surface morphology of 316 steel showed mild deterioration compared to severe surface deterioration, and visible micro/macro-pits on 304L. Despite similar elemental composition, the presence of molybdenum had a strong influence on the corrosion resistance and passivation of 316 steel. 304L steel showed no passivation and resistance to pitting after 0% NaCl concentration coupled with increase in corrosion rate. Results showed that 316 stainless steel significantly performed better than the 304L counterpart with the unusual phenomenon of decreasing corrosion rate with increase in Cl - ion concentration. The corrosion rate, pitting potential, passivation potential and surface morphology of both steel where significantly altered by the Cl - ion concentration, alloy composition and metallurgical properties of the steels. 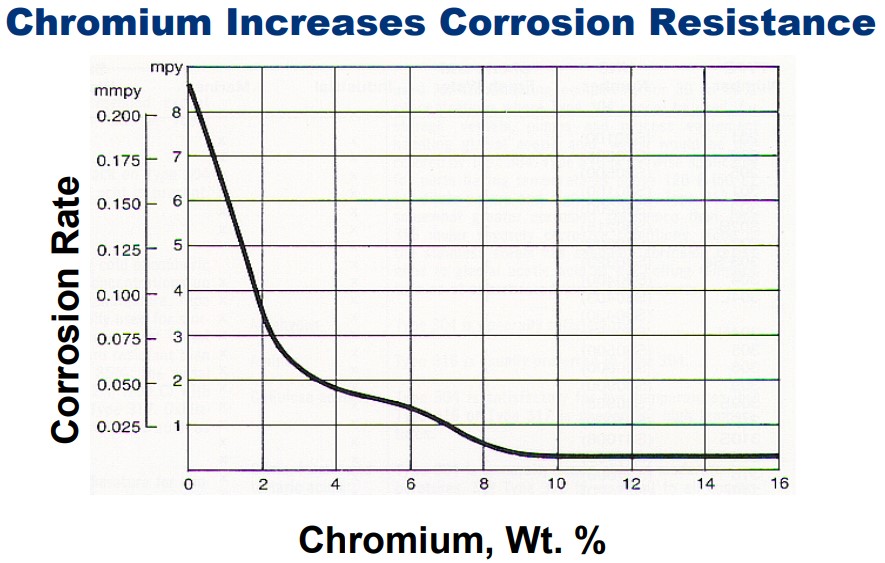
The corrosion resistance of type 304L and 316 austenitic stainless steels in 2M H 2SO 4 at 0-1.5%NaCl concentrations was studied through potentiodynamic polarization technique and optical microscopy analysis. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
